Specifically, chemo-immunotherapy combinations demonstrated particularly encouraging survival outcomes, and among multiple regimens, pembrolizumab + chemotherapy (Pem + chemo) seemed to yield a better survival benefit ( 11, 12). Therefore, combination therapy was explored to improve the efficacy and expand the beneficiaries. Accumulated evidence indicated that a synergistic effect of different regimens contributes to the prolonged survival outcomes ( 9, 10). However, only 20% of patients obtain long-term survival benefit from a single agent of ICIs ( 8). 
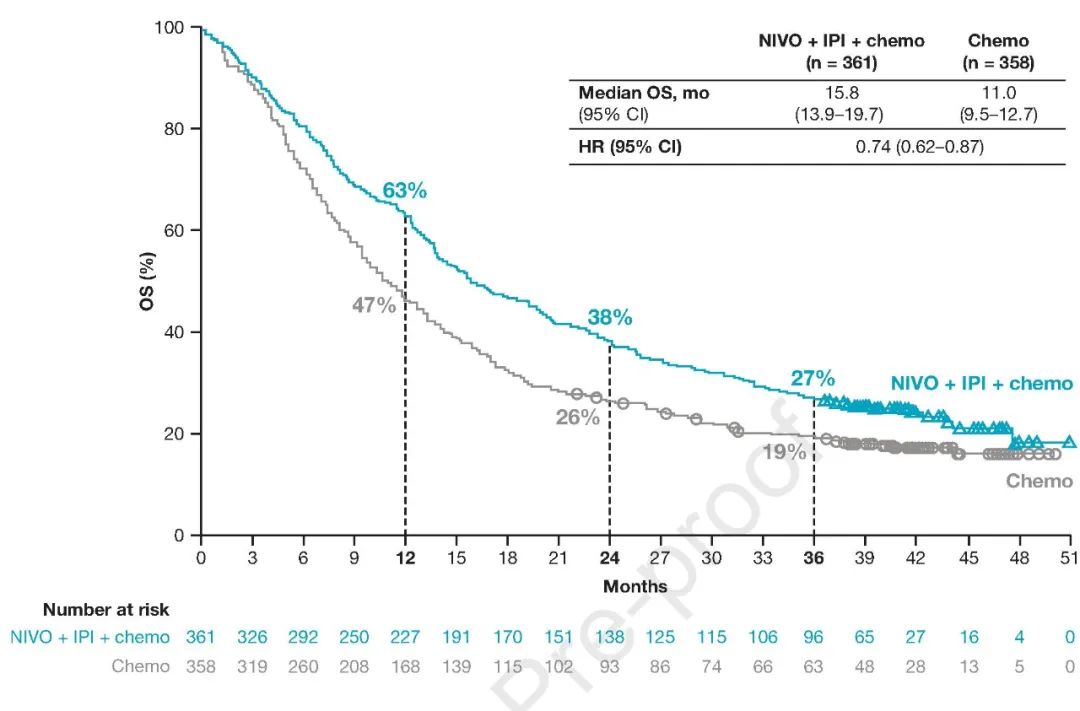
Multiple phase III clinical trials have verified the superior efficacy and acceptable toxicity of ICIs in this population, alone or with other regimens ( 4– 7). Advances in the 1L immunotherapy treatment of driver mutation-negative metastasis non-small cell lung cancer is remarkable ( 3). In the last decade, the treatment landscape of lung cancer has been revolutionized to the era of immunotherapy, and the most prominent representatives are immune checkpoint inhibitors (ICIs), including medications targeting programmed death receptor 1 (PD-1), programmed death-ligand 1 (PD-L1), and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) ( 1, 2). However, for never smoker female patients, Pem + chemo is preferable to choose for demonstrating favorable OS benefit than N-I + chemo. 
As for safety profiles, N-I + chemo showed no significant difference relative to Pem + chemo in any grade adverse events: (RR 1.03, 95% CI 0.99-1.10), but demonstrated reduced toxicity in chemo-related adverse events, such as anemia (RR 0.63, 95% CI 0.49-0.81), neutropenia (RR0.51, 95% CI 0.33-0.79), and thrombocytopenia (RR 0.38, 95% CI 0.21-0.69).Ĭonclusions: N-I + chemo is a promising treatment option for providing comparable OS related to Pem + chemo. The ITC results suggested that N-I + chemo is comparable to Pem + chemo in OS (HR 1.03, 95% CI 0.82-1.30) and ORR (RR 0.81, 95% CI 0.62-1.06), but tended to yield inferior PFS (HR 1.28, 95% CI 1.04-1.59) than did Pem + chemo. Results: Four eligible randomized trials (CheckMate9LA, KEYNOTE-021G, KEYNOTE 189, KEYNOTE 407) involving 2017 patients were available to analyze. Subgroup analyses were stratified according to PD-L1 expression and clinical characteristics. A well-accepted adjusted indirect treatment comparison (ITC) approach was selected to pool efficacy results and safety outcomes. Methods: Relevant databases were searched for eligible trials. We conducted this indirect comparison to compare the efficacy of and safety between these two treatments for providing reference for decision making. 3Centre for Translational Medicine, The First Affiliated Hospital of Xi’an Jiaotong University, Xi’an, Chinaīackground: Nivolumab + ipilimumab + two cycles chemotherapy (N-I + chemo, intensive immunotherapy but chemo-light) and pembrolizumab + chemotherapy (Pem + chemo) were both recommended as first-line treatment for metastatic non-small cell lung carcinoma (NSCLC) patients.2Key Laboratory of Environment and Genes Related to Diseases, Xi’an Jiaotong University, Ministry of Education of China, Xi’an, China.1Department of Medical Oncology, The First Affiliated Hospital of Xi’an Jiaotong University, Xi’an, China.Panpan Jiang 1† Ziyang Mao 1† Qinyang Wang 1 Xiaohui Jia 1 Luying Geng 1 Hong Xu 1 Lili Jiang 1 Chengcheng Yang 1 Min Jiao 1 Hui Guo 1,2,3*
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
